PRODUCTS
- Home
- PRODUCTS
- In Vitro Diagnostics
- Diabetes Care
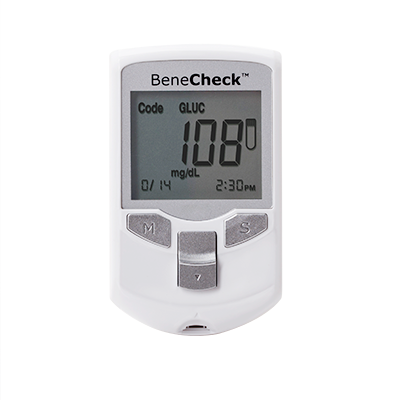
- BeneCheck Supreme Blood Glucose Monitoring System
Diabetes Care
BeneCheck Supreme Blood Glucose Monitoring System
BX-SG00, BX-SG0D- Features
- HCT calibration (broader hematocrit range)
- Bluetooth (BX-SG0D, optional)
- Auto-Ejection
- ISO 13485
- CE certificate
- Made in Taiwan
Description
BeneCheck Supreme Blood Glucose Monitoring System provides accurate blood glucose readings. As a certified medical device with CE approval, it features Hematocrit calibration technology, ensuring precise results by eliminating interference from blood cells across a wide hematocrit range.
Advantages of the BeneCheck Blood Glucose Monitoring System
- Features ultra-wide 0% to 70% Hematocrit (HCT) interference correction technology
- Utilizes No Coding technology; measurement starts immediately upon strip insertion
- Requires only a 0.7 μL micro-sample with rapid results displayed within 5 seconds
- Automatic strip ejection design allows for one-handed disposal after testing
- Employs advanced GDH-FAD (Glucose Dehydrogenase) technology for high interference resistance, unaffected by blood oxygen concentration
- Model BX-SG0D supports Bluetooth connectivity, enabling data transfer to management software for long-term tracking
Blood Glucose Monitoring System FAQ
Test Strip Specifications
| GLU Blood Glucose Strip | |
|---|---|
| Model Number | BK-SG1 |
| Principle | Electrochemistry |
| Test Sample | Fingertip, Venous Blood |
| Hematocrit range | 0% – 70% |
| Measuring Time | 5 Seconds |
| Measuring Range | 20 – 600 mg/dL (1.1 – 33.3 mmol/L) |
| Sample Volume | 0.7 μL |
| Memory | 360 Records |
| Other | GDH-FAD, No Coding |
Device Specifications
Dimensions (L × W × H): Approx. 95 × 60 × 20 mm
Weight: Approx. 71g (including batteries)
Power / Battery: 1.5V AAA Battery × 2